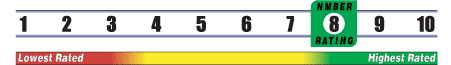
Each tablet contains: Eleutherococcus 600 mg.
Brand name products often contain multiple ingredients. To read detailed information about each ingredient, click on the link for the individual ingredient shown above.
Below is general information about the effectiveness of the known ingredients contained in the product Siberian Ginseng 600 mg. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
INSUFFICIENT RELIABLE EVIDENCE to RATE
Below is general information about the safety of the known ingredients contained in the product Siberian Ginseng 600 mg. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
LIKELY SAFE ...when used orally and appropriately, short-term. Eleuthero root extract 300-2000 mg has been used safely in clinical trials lasting up to 3 months (730,1427,2574,7522,11099,15586,91509). There is insufficient reliable information available about the safety of eleuthero when used long-term.
CHILDREN: POSSIBLY SAFE
when used orally in adolescents aged 12-17 years, short-term.
Eleuthero 750 mg three times daily was used for 6 weeks with apparent safety in one clinical trial (75028). There is insufficient reliable information available about the safety of eleuthero in children or adolescents when used long-term.
PREGNANCY AND LACTATION:
Insufficient reliable information available; avoid using.
Below is general information about the interactions of the known ingredients contained in the product Siberian Ginseng 600 mg. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
Theoretically, eleuthero may have antiplatelet effects and may increase the risk of bleeding if used with anticoagulant or antiplatelet drugs.
|
Theoretically, eleuthero might have additive effects when used with antidiabetes drugs.
Animal research suggests that certain constituents of eleuthero have hypoglycemic activity in both healthy and diabetic animals (7591,73535,74932,74956,74988,74990). A small study in adults with type 2 diabetes also shows that taking eleuthero for 3 months can lower blood glucose levels (91509). However, one very small study in healthy individuals shows that taking powdered eleuthero 3 grams, 40 minutes prior to a 75-gram oral glucose tolerance test, significantly increases postprandial blood glucose levels when compared with placebo (12536). These contradictory findings might be due to patient-specific variability and variability in active ingredient ratios.
|
Theoretically, eleuthero might increase levels of drugs metabolized by CYP1A2.
In vitro and animal research suggest that standardized extracts of eleuthero inhibit CYP1A2 (7532). This effect has not been reported in humans.
|
Theoretically, eleuthero might increase levels of drugs metabolized by CYP2C9.
In vitro and animal research suggest that standardized extracts of eleuthero might inhibit CYP2C9 (7532). This effect has not been reported in humans.
|
Theoretically, eleuthero might increase levels of drugs metabolized by CYP2D6.
|
Theoretically, eleuthero might increase levels of drugs metabolized by CYP3A4.
|
Eleuthero might increase serum digoxin levels and increase the risk of side effects.
In one case report, a 74-year-old male who was stabilized on digoxin presented with an elevated serum digoxin level after starting an eleuthero supplement, without symptoms of toxicity. After stopping the supplement, serum digoxin levels returned to normal (543). It is not clear whether this was due to a pharmacokinetic interaction or to interference with the digoxin assay (15585). Although the product was found to be free of digoxin and digitoxin (543), it was not tested for other contaminants (797).
|
Theoretically, eleuthero might interfere with immunosuppressive drugs because of its immunostimulant activity.
|
Theoretically, eleuthero might decrease levels of drugs metabolized by OATP.
In vitro research suggests that eleuthero inhibits OATP2B1, which might reduce the bioavailability of oral drugs that are substrates of OATP2B1 (35450). Due to the weak inhibitory effect identified in this study, this interaction is not likely to be clinically significant.
|
Theoretically, eleuthero might increase levels of P-glycoprotein substrates.
|
Below is general information about the adverse effects of the known ingredients contained in the product Siberian Ginseng 600 mg. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
General
...Orally, eleuthero root is generally well tolerated when used short-term.
Most Common Adverse Effects:
Orally: Diarrhea, dyspepsia, gastrointestinal upset, headache, nausea, and urticaria.
Cardiovascular ...Orally, increased blood pressure has been reported in children with hypotension taking eleuthero in one clinical study (74980). Eleuthero has been reported to cause tachycardia, hypertension, and pericardial pain in patients with rheumatic heart disease or atherosclerosis. It is unclear if these effects were caused by eleuthero, or by the cardioglycoside-containing herb, silk vine (Periploca sepium), which is a common adulterant found in eleuthero products (12,797,6500).
Dermatologic ...Orally, eleuthero has been reported to cause rash in some clinical studies (75013,75028).
Gastrointestinal ...Orally, eleuthero has been reported to cause dyspepsia, nausea, diarrhea, and gastrointestinal upset in some patients (74938,75028,91510).
Genitourinary ...Orally, mastalgia and uterine bleeding were reported in 7. 3% of females taking eleuthero 2 grams daily in one clinical study (6500,11099). These adverse effects seem to be more likely with higher doses.
Neurologic/CNS
...Orally, headaches have been reported in 9.
8% of people taking eleuthero in one clinical study (11099).
In one case report, a 53-year-old female developed spontaneous subarachnoid hemorrhage associated with the use of an herbal supplement containing red clover, dong quai, and eleuthero (70419). It is unclear if this event was related to the use of eleuthero, the other ingredients, the combination, or another cause entirely.
Psychiatric ...Orally, nervousness has been reported in 7. 3% of people taking eleuthero in one clinical study (11099). Eleuthero has also been reported to cause slight anxiety, irritability, and melancholy in some patients (6500,11099). These adverse effects seem to be more likely to occur with higher doses.
