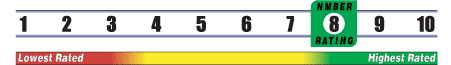
Each capsule contains: Zingiber officinale 550 mg. Other Ingredients: Gelatin, Silicon Dioxide, Sodium Lauryl Sulphate, Water.
Brand name products often contain multiple ingredients. To read detailed information about each ingredient, click on the link for the individual ingredient shown above.

In 2004, Canada began regulating natural medicines as a category of products separate from foods or drugs. These products are officially recognized as "Natural Health Products." These products include vitamins, minerals, herbal preparations, homeopathic products, probiotics, fatty acids, amino acids, and other naturally derived supplements.
In order to be marketed in Canada, natural health products must be licensed. In order to be licensed in Canada, manufacturers must submit applications to Health Canada including information about uses, formulation, dosing, safety, and efficacy.
Products can be licensed based on several criteria. Some products are licensed based on historical or traditional uses. For example, if an herbal product has a history of traditional use, then that product may be acceptable for licensure. In this case, no reliable scientific evidence is required for approval.
For products with non-traditional uses, some level of scientific evidence may be required to support claimed uses. However, a high level of evidence is not necessarily required. Acceptable sources of evidence include at least one well-designed, randomized, controlled trial; well-designed, non-randomized trials; cohort and case control studies; or expert opinion reports.
Finished products licensed by Health Canada must be manufactured according to Good Manufacturing Practices (GMPs) as outlined by Health Canada.
Below is general information about the effectiveness of the known ingredients contained in the product Ginger Root. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
INSUFFICIENT RELIABLE EVIDENCE to RATE
Below is general information about the safety of the known ingredients contained in the product Ginger Root. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
LIKELY SAFE ...when used orally and appropriately. Ginger has been safely used in multiple clinical trials (721,722,723,5343,7048,7084,7085,7400,7623,11346)(12472,13080,13237,13244,17369,17928,17929,89889,89890,89894)(89895,89898,89899,90102,96252,96253,96259,96260,96669) (101760,101761,101762,103359,107903).
POSSIBLY SAFE ...when used topically and appropriately, short-term (89893,89897).
CHILDREN: LIKELY SAFE
when consumed in the amounts typically found in foods.
CHILDREN: POSSIBLY SAFE
when used orally and appropriately, short-term.
Ginger powder has been used with apparent safety at a dose of up to 750 mg daily for 4 days in girls aged 14-18 years (96255).
PREGNANCY: LIKELY SAFE
when consumed in the amounts typically found in foods.
Ginger is considered a first-line nonpharmacological treatment option for nausea in pregnancy by the American College of Obstetrics and Gynecology (ACOG) (111601). However, it should not be used long-term or without medical supervision and close monitoring.
PREGNANCY: POSSIBLY SAFE
when used for medicinal purposes.
Despite some early reports of adverse effects (721,7083) and one observational study suggesting that taking dried ginger and other herbal supplements during the first 20 weeks of pregnancy marginally increased the chance of stillbirth (96254), most research shows that ginger is unlikely to cause harm to the baby. The risk for major malformations in infants of parents who took ginger when pregnant does not appear to be higher than the baseline rate of 1% to 3% (721,1922,5343,11346,13071,13080,96254). Also, other research suggests that ginger intake during various trimesters does not significantly affect the risk of spontaneous abortion, congenital malformations, stillbirth, perinatal death, preterm birth, low birth weight, or low Apgar scores (18211,90103). Ginger use has been associated with an increase in non-severe vaginal bleeding, including spotting, after week 17 of pregnancy (18211).
LACTATION: LIKELY SAFE
when consumed in the amounts typically found in foods.
There is insufficient reliable information available about the safety of ginger when used for medicinal purposes; avoid amounts greater than those found in foods.
Below is general information about the interactions of the known ingredients contained in the product Ginger Root. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
Ginger may have antiplatelet effects and may increase the risk of bleeding if used with anticoagulant or antiplatelet drugs. However, research is conflicting.
Laboratory research suggests that ginger inhibits thromboxane synthetase and decreases platelet aggregation (7622,12634,20321,20322,20323,96257). However, this has not been demonstrated unequivocally in humans, with mixed results from clinical trials (96257). Theoretically, excessive amounts of ginger might increase the risk of bleeding when used with anticoagulant/antiplatelet drugs.
|
Theoretically, taking ginger with antidiabetes drugs might increase the risk of hypoglycemia.
|
Theoretically, taking ginger with calcium channel blockers might increase the risk of hypotension.
Some animal and in vitro research suggests that ginger has hypotensive and calcium channel-blocking effects (12633). Another animal study shows that concomitant administration of ginger and the calcium channel blocker amlodipine leads to greater reductions in blood pressure when compared with amlodipine alone (107901).
|
Theoretically, when taken prior to cyclosporine, ginger might decrease cyclosporine levels.
In an animal model, ginger juice taken 2 hours prior to cyclosporine administration reduced the maximum concentration and area under the curve of cyclosporine by 51% and 40%, respectively. This effect was not observed when ginger juice and cyclosporine were administered at the same time (20401).
|
Theoretically, ginger might increase the levels of CYP1A2 substrates.
In vitro research shows that ginger inhibits CYP1A2 activity (111544). However, this interaction has not been reported in humans.
|
Theoretically, ginger might increase the levels of CYP2B6 substrates.
In vitro research shows that ginger inhibits CYP2B6 activity (111544). However, this interaction has not been reported in humans.
|
Theoretically, ginger might increase the levels of CYP2C9 substrates.
In vitro research shows that ginger inhibits CYP2C9 activity (111544). However, this interaction has not been reported in humans.
|
Ginger might increase or decrease the levels of CYP3A4 substrates.
In vitro research and some case reports suggest that ginger inhibits CYP3A4 activity (111544,111644). Three case reports from the World Health Organization (WHO) adverse drug reaction database describe increased toxicity in patients taking ginger and cancer medications that are CYP3A4 substrates (imatinib, dabrafenib, and crizotinib). However, the causality of this interaction is unclear due to the presence of multiple interacting drugs and routes of administration (111644).
Conversely, other in vitro research suggests that ginger induces CYP3A4 activity, leading to reduced levels of CYP3A4 substrates (111404). However, this interaction has not been reported in humans. |
Theoretically, ginger might increase levels of losartan and the risk of hypotension.
In animal research, ginger increased the levels and hypotensive effects of a single dose of losartan (102459). It is not clear if ginger alters the concentration or effects of losartan when taken continuously. Additionally, this interaction has not been shown in humans.
|
Theoretically, ginger might increase levels of metronidazole.
In an animal model, ginger increased the absorption and plasma half-life of metronidazole. In addition, the elimination rate and clearance of metronidazole was significantly reduced (20350).
|
Ginger may have antiplatelet effects and increase the risk of bleeding if used with nifedipine.
Clinical research shows that combined treatment with ginger 1 gram plus nifedipine 10 mg significantly inhibits platelet aggregation when compared to nifedipine or ginger alone (20324).
|
Ginger might increase the absorption and blood levels of P-glycoprotein (P-gp) substrates.
In vitro research and case reports suggest that ginger inhibits drug efflux by P-gp, potentially increasing absorption and serum levels of P-gp substrates (111544,111644). Two case reports from the World Health Organization (WHO) adverse drug reaction database describe increased toxicity in patients taking ginger and cancer medications that are P-gp substrates (trametinib, crizotinib). However, the causality of this interaction is unclear due to the presence of multiple interacting drugs and routes of administration (111644).
|
Ginger might increase the risk of bleeding with phenprocoumon.
Phenprocoumon, a warfarin-related anticoagulant, might increase the international normalized ratio (INR) when taken with ginger. There is one case report of a 76-year-old woman with a stable INR on phenprocoumon that increased to greater than 10 when she began consuming dried ginger and ginger tea (12880).
|
Ginger might increase the risk of bleeding with warfarin.
Laboratory research suggests that ginger might inhibit thromboxane synthetase and decrease platelet aggregation (7622,12634,20321,20322,20323). In one case report, ginger increased the INR when taken with phenprocoumon, which has similar pharmacological effects as warfarin (12880). In another case report, ginger increased the INR when taken with a combination of warfarin, hydrochlorothiazide, and acetaminophen (20349). A longitudinal analysis suggests that taking ginger increases the risk of bleeding in patients taking warfarin for at least 4 months (20348). However, research in healthy people suggests that ginger has no effect on INR, or the pharmacokinetics or pharmacodynamics of warfarin (12881,15176). Until more is known, monitor INRs closely in patients taking large amounts of ginger.
|
Below is general information about the adverse effects of the known ingredients contained in the product Ginger Root. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
General
...Orally, ginger is generally well tolerated.
However, higher doses of 5 grams per day increase the risk of side effects and reduce tolerability. Topically, ginger seems to be well tolerated.
Most Common Adverse Effects:
Orally: Abdominal discomfort, burping, diarrhea, heartburn, and a pepper-like irritant effect in the mouth and throat. However, some of these mild symptoms may be reduced by ingesting encapsulated ginger in place of powdered ginger.
Topically: Dermatitis in sensitive individuals.
Cardiovascular ...Orally, use of ginger resulted in mild arrhythmia in one patient in a clinical trial (16306).
Dermatologic
...Orally, ginger can cause hives (17933), as well as bruising and flushing (20316) or rash (20316).
Topically, ginger can cause dermatitis in sensitive individuals (12635,46902).
Gastrointestinal
...Orally, common side effects of ginger include nausea (17933,22602,89898,101761), belching (10380,103359), dry mouth (103359), dry retching (10380), vomiting (10380), burning sensation (10380), oral numbness (22602), abdominal discomfort (5343,89898,96253), heartburn (5343,7624,12472,16306,20316,51845,89894,89895,89898,89899)(101760,101761,101762,111543), diarrhea (5343,101760), constipation (89898,101760,101761), or a transient burning or "chilly hot" sensation of the tongue and throat (52076).
Orally, Number Ten, a specific product composed of rhubarb, ginger, astragalus, red sage, and turmeric, can increase the incidence of loose stools (20346).
Four cases of small bowel obstruction due to ginger bolus have been reported following the ingestion of raw ginger without sufficient mastication (chewing). In each case, the bolus was removed by enterotomy. Ginger is composed of cellulose and therefore is resistant to digestion. It can absorb water, which may cause it to swell and become lodged in narrow areas of the digestive tract (52115).
Genitourinary ...In one clinical trial, some patients reported increased menstrual bleeding while taking a specific ginger extract (Zintoma, Goldaru) 250 mg four times daily orally for 3 days (17931). An "intense" urge to urinate after 30 minutes was reported in two of eight patients given 0.5-1 gram of ginger (7624). However, this effect has not been corroborated elsewhere. Dysuria, flank pain, perineal pain, and urinary stream interruption have been reported in a 43-year-old male who drank ginger tea, containing 2-3 teaspoons of dry ginger, daily over 15 years. The adverse effects persisted for 4 years and were not associated with increases in urinary frequency or urgency. Upon discontinuing ginger, the patient's symptoms began to improve within one week and completely resolved after eight weeks, with no relapses six months later (107902).
Immunologic ...In one case report, a 59-year-old Japanese female with multiple allergic sensitivities developed pruritus and then anaphylactic shock after taking an oral ginger-containing herbal supplement for motion sickness (Keimei Gashinsan, Keimeido). The patient had used this supplement previously for over 20 years with no allergic reaction. The authors theorized the development of a cross-reactivity to ginger after the use of an oral supplement containing zedoary and turmeric, which are also in the Zingiberaceae family (102463).
Neurologic/CNS ...Orally, ginger may cause sedation, drowsiness, or dizziness (16306,17933,51845).
