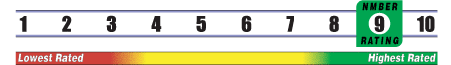
Each capsule contains: Plantago Ovata 520 mg. Other Ingredients: Gelatin, Magnesium Stearate.
Brand name products often contain multiple ingredients. To read detailed information about each ingredient, click on the link for the individual ingredient shown above.

In 2004, Canada began regulating natural medicines as a category of products separate from foods or drugs. These products are officially recognized as "Natural Health Products." These products include vitamins, minerals, herbal preparations, homeopathic products, probiotics, fatty acids, amino acids, and other naturally derived supplements.
In order to be marketed in Canada, natural health products must be licensed. In order to be licensed in Canada, manufacturers must submit applications to Health Canada including information about uses, formulation, dosing, safety, and efficacy.
Products can be licensed based on several criteria. Some products are licensed based on historical or traditional uses. For example, if an herbal product has a history of traditional use, then that product may be acceptable for licensure. In this case, no reliable scientific evidence is required for approval.
For products with non-traditional uses, some level of scientific evidence may be required to support claimed uses. However, a high level of evidence is not necessarily required. Acceptable sources of evidence include at least one well-designed, randomized, controlled trial; well-designed, non-randomized trials; cohort and case control studies; or expert opinion reports.
Finished products licensed by Health Canada must be manufactured according to Good Manufacturing Practices (GMPs) as outlined by Health Canada.
Below is general information about the effectiveness of the known ingredients contained in the product Psyllium Seed Husks 520 mg Capsules. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
INSUFFICIENT RELIABLE EVIDENCE to RATE
Below is general information about the safety of the known ingredients contained in the product Psyllium Seed Husks 520 mg Capsules. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
LIKELY SAFE ...when used orally with appropriate fluid intake (93216). Blond psyllium preparations have been safely used in doses up to 20 grams per day for up to 6 months (1376,2324,2327,6261,6262,8060,8061,8066,8423,9422) (10095,13102,22961,22962,22963,22964,22966,54260,22968,22969) (22970,22972,22973,22976,22977,22978,22979,22980,22981,22986) (22987,22988,22989,22990,22992,22993,22994,22995,22996,22998) (23402,23403,23404,23405,92198,106859). The U.S. Food and Drug Administration (FDA) requires over-the-counter medicines that contain dry or incompletely hydrated psyllium to carry a warning that they should be taken with at a least a full glass of liquid to reduce the risk of choking. This labeling also applies to foods containing psyllium that are marketed with a health claim regarding coronary heart disease (93217,93218).
POSSIBLY SAFE ...when used in eye drops. Blond psyllium mucilage has been used with apparent safety in eye drops four times daily for 6 weeks (105274). There is insufficient reliable information available about the safety of blond psyllium when used topically.
LIKELY UNSAFE ...when used orally without adequate fluid intake due to the risk for choking and gastrointestinal obstruction (93218). ...when granular dosage forms containing blond psyllium are used as over the counter (OTC) laxatives. The U.S. Food and Drug Administration (FDA) states that these granular dosage forms are not generally recognized as safe and effective as OTC laxatives due to an increased risk of choking and gastrointestinal obstruction (93219).
CHILDREN: POSSIBLY SAFE
when used orally and appropriately, short-term.
Blond psyllium husk has been used with apparent safety in doses up to 12 grams daily for 4 weeks (110763).
PREGNANCY AND LACTATION: LIKELY SAFE
when used orally and appropriately (272).
Below is general information about the interactions of the known ingredients contained in the product Psyllium Seed Husks 520 mg Capsules. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
Theoretically, blond psyllium might reduce the effects of carbamazepine and increase the risk for convulsions.
|
Theoretically, taking blond psyllium at the same time as digoxin might reduce digoxin absorption.
|
Theoretically, taking blond psyllium at the same time as ethinyl estradiol might alter levels of estradiol.
Concurrent use of blond psyllium with ethinyl estradiol results in a slight increase in the extent of ethinyl estradiol absorption and a slower rate of absorption. However, this is unlikely to be clinically significant (12421).
|
Theoretically, taking blond psyllium at the same time as lithium might reduce lithium absorption.
|
Theoretically, blond psyllium might increase the therapeutic and adverse effects of metformin.
Concurrent use of blond psyllium with metformin slows and increases metformin absorption (99433). To avoid changes in absorption, take psyllium 30-60 minutes after metformin.
|
Theoretically, taking blond psyllium at the same time as olanzapine might reduce olanzapine absorption.
The fiber in blond psyllium might decrease the absorption of olanzapine. A single case report describes a reduction in the effectiveness of olanzapine when it was taken concomitantly with an unspecified type of psyllium 3 grams orally twice daily. This effect was reversed when psyllium was stopped (106858).
|
Theoretically, psyllium might increase, decrease, or have no effect on the absorption of oral drugs.
Psyllium seems to have variable effects on drug absorption. To avoid changes in absorption, take psyllium 30-60 minutes after oral medications. Animal research shows that blond psyllium delays and increases the absorption of metformin and ethinyl estradiol (12421,99433). Conversely, case reports and animal research suggest that blond psyllium might reduce absorption of lithium, digoxin, olanzapine, and carbamazepine (12,18,272,93214,106858). Finally, some pharmacokinetic studies show that psyllium does not affect the absorption of levothyroxine or warfarin (12420,103940).
|
Below is general information about the adverse effects of the known ingredients contained in the product Psyllium Seed Husks 520 mg Capsules. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
General
...Orally, blond psyllium is generally well tolerated.
When used as eye drops, blond psyllium seems to be well tolerated.
Most Common Adverse Effects:
Oral: Abdominal pain, constipation, diarrhea, dyspepsia, flatulence, and nausea.
Serious Adverse Effects (Rare):
Oral: Bowel obstruction, esophageal obstruction.
Gastrointestinal ...Orally, blond psyllium can cause transient flatulence, abdominal pain, diarrhea, constipation, dyspepsia, and nausea (1376). Starting with a low dose and slowly titrating to the desired dose can often minimize gastrointestinal side effects. There is some concern that blond psyllium can cause esophageal or bowel obstruction when consumed without water or in patients with swallowing disorders (604,8080,8081,110760). Tell patients to consume plenty of water when taking blond psyllium. Suggest at least 240 mL of fluid for every 3.5-5 grams of seed husk or 7 grams of seed (1376,8080,8081).
Musculoskeletal ...Orally, backache has been reported with the use of psyllium (1376).
Neurologic/CNS ...Orally, headache has been reported with the use of psyllium (1376).
Ocular/Otic ...Ophthalmically, blurred vision or burning haven been reported rarely in patients using eye drops containing blond psyllium mucilage (105274).
Pulmonary/Respiratory ...Orally, rhinitis, increased cough, and sinusitis have been reported with the use of psyllium (1376).
Other
...Some patients can have an allergic response to blond psyllium.
Allergy symptoms include allergic rhinitis, sneezing, conjunctivitis, urticarial rash, itching, flushing, and dyspnea. More serious symptoms include wheezing, facial and body swelling, chest congestion, chest and throat tightness, cough, diarrhea, hypotension, loss of consciousness, and anaphylactic shock. Occupational exposure or repeated ingestion of psyllium can cause sensitization, which can lead to serious allergic reactions (2328,2329,2330,8079,9246,92193). Severe allergic reactions may occur after eating a small quantity of cereal that contains blond psyllium. At least one cereal (Heartwise, Kellogg Co.) has increased the purity of the psyllium it contains, which has decreased the incidence of allergic reactions (9244). A warning of the potential for allergic reactions is on the label of all cereals that contain psyllium (9247). Patients hypersensitive to psyllium usually have marked eosinophilia and an elevated psyllium-specific IgE antibody serum level (2328,2329,92193).
There is concern that individuals allergic to pollen from English plantain weed (Plantain lanceolate) might also react to psyllium husk dust; however, it appears that there is little cross-allergenicity between these plants and is probably mild and of no clinical significance (8057,9244,92193).
Blond psyllium has a tendency to plug feeding tubes. This can be avoided if blond psyllium is mixed with water and pushed through the feeding tube in less than 5 minutes (8423).
