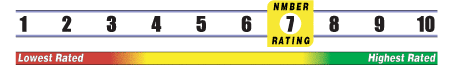
| Ingredients | Amount Per Serving |
|---|---|
|
(Nigella sativa )
(seed)
|
500 mg |
Gelatin, Glycerin, purified Water
Below is general information about the effectiveness of the known ingredients contained in the product Black Cumin Seed Oil Organic. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
INSUFFICIENT RELIABLE EVIDENCE to RATE
Below is general information about the safety of the known ingredients contained in the product Black Cumin Seed Oil Organic. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
LIKELY SAFE ...when used orally in amounts found in foods (6).
POSSIBLY SAFE ...when black seed oil is used orally at doses of up to 5 mL or 5 grams daily for up to 12 weeks (36071,94486,96927,96929,98815,101550,102062,110269,110276). ...when a specific black seed oil formulation containing 5% thymoquinone (BlaQmax) is used in doses of 200 mg daily for 90 days (110264). ...when black seed powder is used orally at doses of up to 2 grams daily for up to 12 months (36239,36244,94478,94479,94485,96928,102061,110268,110271) or 3 grams daily for 3 months (114880). ...when used topically and appropriately, short-term. There is some clinical evidence that black seed oil can be safely applied as an oil 2 times daily for up to 6 months or 3 times daily for up to 1 month (95981,98814,102064,110262), as a 30% gel twice daily for approximately 2 months (94483), or as a 10% solution in a patch to oral lesions 3 times daily for 4 weeks (114815).
CHILDREN: POSSIBLY SAFE
when black seed oil is used orally at doses of 40-80 mg/kg daily for 2-19 months in children ages 4-17 years old (36071,95984).
However, the higher dose of 80 mg/kg daily has been associated with increased adverse effects such as gastrointestinal complaints when taken on an empty stomach (36071).
PREGNANCY: LIKELY UNSAFE
when used orally in amounts exceeding those found in food.
Black seed may decrease or inhibit uterine contractions (241) and may have contraceptive activity (242).
LACTATION:
Insufficient reliable information available; avoid using.
Below is general information about the interactions of the known ingredients contained in the product Black Cumin Seed Oil Organic. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
Theoretically, black seed may increase the risk of bleeding if used with anticoagulant or antiplatelet drugs.
|
Theoretically, taking black seed with antidiabetes drugs might increase the risk of hypoglycemia.
|
Theoretically, taking black seed with antihypertensive drugs might increase the risk of hypotension.
Clinical research suggests that black seed powder and oil might reduce blood pressure by 2-3 mmHg (16437,94489). In animal research, black seed modestly reduces blood pressure and concomitant use of black seed and amlodipine (Norvasc) or metoprolol (Lopressor) increased the blood pressure lowering effects of these drugs (101559,108703).
|
Theoretically, black seed may increase the risk of bleeding if used with clopidogrel.
Animal research shows that taking black seed extract daily for 2 weeks prior to a single dose of clopidogrel increases maximum concentrations of clopidogrel by approximately 31% and modestly decreases oral clearance. Furthermore, bleeding time was increased by 12% (108701). This has not been shown in humans.
|
Theoretically, concomitant use with drugs that have sedative properties may cause additive effects.
Animal research suggests that black seed may have CNS depressant effects (36064).
|
Theoretically taking black seed might reduce the levels and clinical effects of cyclosporine.
In animal research, black seed extract decreased the maximal levels of cyclosporine in the blood by 35.5% (94474). This has not been shown in humans.
|
Theoretically, black seed might increase levels of drugs metabolized by CYP2C9.
In vitro research suggests that thymoquinone, a constituent of black seed, can decrease the metabolism of phenytoin by a mechanism possibly related to the inhibition of CYP2C9 (110281). The effect of black seed on CYP2C9 is unclear. This has not been shown in humans.
|
Theoretically, taking black seed with diuretic drugs might increase potassium loss and the risk of hypokalemia.
Black seed extract has shown diuretic effects in animals, which could theoretically increase potassium loss (36026). This has not been shown in humans.
|
Theoretically, black seed might interfere with immunosuppressive therapy.
|
Theoretically, black seed might increase or decrease levels and effects of phenytoin.
In vitro research suggests that thymoquinone, a constituent of black seed, can decrease the metabolism of phenytoin (110281). This effect may be due to inhibition of cytochrome P450 2C9 (CYP2C9). However, animal research shows that black seed decreases the maximum concentration of and total systemic exposure to phenytoin by 57% and 87%, respectively. This seems to be related to increased clearance and steady state volume of distribution (110905). This interaction has not been shown in humans.
|
Theoretically black seed might reduce plasma levels and therapeutic effects of prednisolone.
In animal research, oral administration of a single dose of black seed oil 15 minutes prior to oral prednisolone decreases the prednisolone maximum plasma concentration by 65% and area under the curve by 25%(114811). This has not been shown in humans.
|
Theoretically, combining serotonergic drugs with black seed might increase the risk of serotonergic side effects, including serotonin syndrome and cerebral vasoconstrictive disorders.
Animal research suggests that black seed can increase brain serotonin levels (36180,94488). In one case report, a 35-year-old man undergoing endoscopic surgery experienced immediate postoperative serotonin syndrome that was likely associated with the use of black seed oil 600 mg daily starting 4 days before surgery, and precipitated by the use of serotonergic pain medications, including fentanyl and oxycodone (101558). Monitor patients for signs of serotonin syndrome and other serotonergic side effects if using black seed with serotonergic drugs.
|
Theoretically, black seed might reduce plasma levels and the therapeutic effects of sildenafil.
Animal research shows that black seed reduces the total systemic exposure to sildenafil by 43% (110898). So far, this interaction has not been reported in humans.
|
Theoretically, black seed might increase levels of warfarin and increase the risk of bleeding.
In vitro research suggests that thymoquinone, a constituent of black seed, can decrease the metabolism of warfarin (110280). This effect may be due to inhibition of cytochrome P450 2C9 (CYP2C9). The effect of black seed on warfarin metabolism is unclear. This has not been shown in humans.
|
Below is general information about the adverse effects of the known ingredients contained in the product Black Cumin Seed Oil Organic. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
General
...Orally and topically, black seed seems to be well tolerated.
Most Common Adverse Effects:
Orally: Constipation, gastric burning or discomfort, nausea, and vomiting.
Dermatologic ...Orally, black seed can cause itching, but this adverse effect is rare (94481). It has also been reported to cause bullous drug eruption and maculopapular eruption (94480,100324). Topically, black seed and its oil have been reported to cause allergic contact dermatitis (36053,36296,36297,100324). Rarely, topical use of the oil has been reported to cause a rash related to drug reaction with eosinophilia and systemic symptoms (DRESS) (110277).
Gastrointestinal ...In a clinical trial, unspecified gastrointestinal complaints occurred in 20% of patients taking black seed oil orally (36071). Gastrointestinal complaints occurred rarely in another clinical trial; however, one patient in this study was hospitalized for nausea and vomiting thought to be associated with black seed oil (110267). Gastrointestinal adverse effects to black seed have included constipation (36180), burning sensation (94487), epigastric discomfort (94479), vomiting (94491), or mild nausea (94481,94489,94491,96927,96929). Gastrointestinal adverse effects to a specific black seed oil formulation containing 5% thymoquinone (BlaQmax) have included mild cases of bloating, burping and mild diarrhea (110264).
Genitourinary ...Orally, relatively severe menstrual bleeding has occurred in one clinical trial (104661).
Hepatic ...While intake of black seed has been associated with hepatotoxicity in some animal research, other animal research has not confirmed this finding (245,95982). One case report describes hepatotoxicity, kidney injury, and rhabdomyolysis occurring in an otherwise healthy 26-year-old male after taking black seed oil 1000 mg twice daily for 1 month. The patient was hospitalized for critical care management, including hemodialysis. After 52 days in the hospital, the patient had recovered and was discharged (114812).
Immunologic ...Orally, black seed has been reported to cause bullous drug eruption and maculopapular eruption (94480,100324). Topically, black seed and its oil can cause allergic contact dermatitis (6,36053,36296,36297,100324,110266). One case of bullous drug eruption with skin detachment has been reported for a 54-year-old woman who used black seed oil orally. The eruptions resolved following treatment with clobetasol propionate 0.05%. Although this patient showed a positive skin prick test for undiluted black seed oil, the tests were negative when the oil was diluted to 10% and 1% (94480). A 28-year-old woman developed a rash following topical use of black seed oil. She was diagnosed with drug reaction with eosinophilia and systemic symptoms (DRESS), including enlarged lymph nodes, and required systemic corticosteroid treatment. This diagnosis was confirmed six months later following confirmation with a patch test (110277). In another case report, a 58-year-old woman developed eczematous lesions on the lower and upper eyelids after topical application of an oil containing black seed. This reaction was followed by the development of a diffuse maculopapular eruption after taking two oral capsules containing black seed oil. It is theorized that the topical application of black seed oil led to systemic sensitization prior to the use of oral black seed in this patient (100324).
Renal ...Orally, black seed might cause renal dysfunction. A case of acute renal failure thought to be related to use of black seed tablets 2-2.5 grams daily has been reported for a 62 year-old patient with diabetes (94477). Additionally, a case of acute kidney injury, rhabdomyolysis, and hepatotoxicity has been reported in an otherwise healthy 26-year-old male after taking black seed oil 1000 mg twice daily for 1 month. The patient was hospitalized for critical care management, including hemodialysis. . After 52 days in the hospital, the patient had recovered and was discharged (114812).
