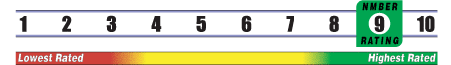
| Ingredients | Information For Compounding |
|---|---|
|
(Betaine Anhydrous)
|
4.9 Gram(s) |
Below is general information about the effectiveness of the known ingredients contained in the product TMG (Trimethylglycine). Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
INSUFFICIENT RELIABLE EVIDENCE to RATE
Below is general information about the safety of the known ingredients contained in the product TMG (Trimethylglycine). Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
LIKELY SAFE ...when used orally and appropriately in doses of up to 6 grams daily (698,10631). However, some patients have used up to 20 grams daily with apparent safety (698). Betaine anhydrous is available as an FDA-approved prescription product (Cystadane) (698), and also as a supplement. The European Food Safety Authority states that betaine anhydrous is safe to use in doses up to 6 mg/kg daily, in addition to usual dietary intake (105548). There is insufficient reliable information available about the safety of topical betaine anhydrous.
CHILDREN: LIKELY SAFE
when used orally and appropriately in doses up to 150 mg/kg daily (698).
However, some patients have used up to 20 grams daily with apparent safety (698). Prescription betaine anhydrous (Cystadane) is approved by the US FDA for use in infants and children (698).
PREGNANCY AND LACTATION:
Insufficient reliable information available; avoid using.
Below is general information about the interactions of the known ingredients contained in the product TMG (Trimethylglycine). Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
Below is general information about the adverse effects of the known ingredients contained in the product TMG (Trimethylglycine). Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
General
...Orally, betaine anhydrous is generally well tolerated.
Most Common Adverse Effects:
Orally: Body odor, diarrhea, elevated cholesterol levels, GI distress, nausea, vomiting.
Serious Adverse Effects (Rare):
Orally: Cerebral edema.
Cardiovascular ...Betaine anhydrous might have adverse effects on the plasma lipid profile. Some studies have reported a 3% to 4% increase in total and low-density lipoprotein (LDL) cholesterol levels with betaine anhydrous 6 grams daily (16452,16455,16456,34904). A meta-analysis of 6 studies in adults, some with obesity and/or prediabetes, shows that taking betaine anhydrous 4-6 grams daily for 6-24 weeks is associated with a mean increase in total cholesterol of 4 mg/dL, with no significant change in LDL cholesterol, high-density lipoprotein (HDL) cholesterol, or triglyceride levels (105814). Another meta-analysis of 12 studies, some in healthy adults and others in adults with various disease states, shows that taking betaine anhydrous 1.5-20 grams daily for 2-52 weeks is associated with a mean increase in total cholesterol of 14 mg/dL, and a mean increase in LDL cholesterol of 10 mg/dL, with no change in triglyceride or HDL cholesterol levels (105813).
Gastrointestinal ...Orally, betaine anhydrous can cause vomiting, nausea, GI distress, and diarrhea (698,10631,34888,34928,111374).
Neurologic/CNS ...When used orally to treat homocystinuria due to cystathionine beta-synthase deficiency, elevated plasma methionine concentrations can occur following use of betaine anhydrous, which might lead to cerebral edema (698,111374).
Other ...Orally, betaine anhydrous can cause body odor (698,10631).
