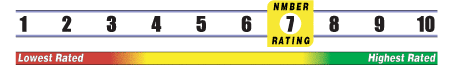
| Ingredients | Amount Per Serving |
|---|---|
|
(Melissa officinalis )
(aerial)
(5% Rosmarinic Acid)
(Lemon Balm extract (Form: 5% Rosmarinic Acid) PlantPart: aerial Genus: Melissa Species: officinalis )
|
1000 mg |
Vegetarian Capsules (Form: Vegetable Cellulose), Maltodextrin, Nu-FLOW Rice Fiber Note: (non-GMO) (less than or equal to 3%)
Below is general information about the effectiveness of the known ingredients contained in the product Lemon Balm Extract 500 mg. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
INSUFFICIENT RELIABLE EVIDENCE to RATE
Below is general information about the safety of the known ingredients contained in the product Lemon Balm Extract 500 mg. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
LIKELY SAFE ...when used orally in amounts commonly found in foods. Lemon balm has Generally Recognized as Safe (GRAS) status in the US (4912).
POSSIBLY SAFE ...when used orally and appropriately, short-term. Lemon balm extract has been used with apparent safety at a dose of 500 mg daily for 6 months or at a dose of 3000 mg daily for 2 months (9993,9994,104435,104435,110136). ...when used topically and appropriately, short-term. Lemon balm 1% dried leaf extract has been used up to 4 times daily with apparent safety for a few days (790,9995).
CHILDREN: POSSIBLY SAFE
when used orally and appropriate, short-term.
A single dose of lemon balm extract 3-6 mg/kg has been safely used in children aged 6-7 years (19525). A specific combination product providing lemon balm leaf extract 80 mg and valerian root extract 160 mg (Euvegal forte, Dr. Willmar Schwabe Pharmaceuticals) 1-2 tablets once or twice daily has been safely used in children under 12 years of age for 30 days (14416). In infants up to 4 weeks old, multi-ingredient products (ColiMil, ColiMil Plus) containing lemon balm 64-97 mg daily have been used with apparent safety for up to 7 days (16735,96278).
PREGNANCY AND LACTATION:
Insufficient reliable information available; avoid using.
Below is general information about the interactions of the known ingredients contained in the product Lemon Balm Extract 500 mg. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
Theoretically, concomitant use of lemon balm might have additive effects with CNS depressant drugs.
|
Theoretically, lemon balm might interfere with thyroid hormone replacement therapy.
In vitro, constituents of lemon balm extract bind to thyroid stimulating hormone (TSH), preventing TSH receptor-binding and leading to the inhibition of TSH-stimulated adenylate cyclase activity (19727,19728). In animals, lemon balm extract has been shown to decrease levels of circulating TSH and inhibit thyroid secretion (19726).
|
Below is general information about the adverse effects of the known ingredients contained in the product Lemon Balm Extract 500 mg. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
General
...Orally, lemon balm seems to be well tolerated in food amounts and larger, medicinal amounts.
Topically, lemon balm seems to be well tolerated.
Serious Adverse Effects (Rare):
Orally: Wheezing has been rarely reported.
Cardiovascular ...Orally, a case of transient complete atrioventricular block and QT prolongation is reported in a 25-year-old female following the post-workout use of a specific product (Muscle Eze Advanced) containing lemon balm and several other ingredients. Symptoms of fatigue and lightheadedness started 1 week into use of the product. Product discontinuation led to restoration of normal sinus rhythm within 24 hours and normalization of the electrocardiogram within 2 weeks (112556). It is unclear whether this occurrence is due to lemon balm, other ingredients, or the combination.
Dermatologic ...Topically, lemon balm 1% cream applied 5 times daily to cold sores has been associated with two cases of irritation and one case of cold sore exacerbation. However, these effects do not appear to occur more often with lemon balm than with placebo (790).
Gastrointestinal ...Orally, lemon balm might increase appetite in some patients (91732,104433). Nausea, vomiting, and abdominal pain have been reported rarely and do not seem to occur more often than in patients taking placebo (9993).
Neurologic/CNS ...Orally, lemon balm has been reported to cause dizziness and sedation; however, it does not seem to occur more often with lemon balm than placebo (9993,104433). Additionally, other clinical research shows that using lemon balm in conjunction with alcohol does not affect reaction time or influence cognitive performance (19427,19723).
Pulmonary/Respiratory ...Orally, lemon balm has been associated with rare cases of wheezing (9993).
