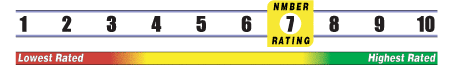
| Ingredients | Not Present |
|---|---|
|
(Sanguinaria canadensis )
(root)
(wildcrafted, dry herb strength: 1:4 (250 mg/ml))
|
Alcohol, Water, Filtered
Below is general information about the effectiveness of the known ingredients contained in the product Blood Root. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
INSUFFICIENT RELIABLE EVIDENCE to RATE
INSUFFICIENT RELIABLE EVIDENCE to RATE
Below is general information about the safety of the known ingredients contained in the product Blood Root. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
POSSIBLY SAFE ...when used orally and appropriately short-term (4).
POSSIBLY UNSAFE ...when excessive doses are used orally. The bloodroot constituent sanguinarine, although thought to be poorly absorbed, is a toxic alkaloid (6,12). ...when applied topically. Use of toothpaste or mouthwash containing bloodroot has been associated with an increased risk of developing oral leukoplakia (36666,36668). When applied to the skin, bloodroot paste causes pain, skin erosion, and a thick scab called an eschar which falls off leaving an indented scar. The U.S. Food and Drug Administration recommends that patients avoid bloodroot containing topical products such as "black salve" (53499,95442,95444,95445,95446).
PREGNANCY: LIKELY UNSAFE
when used orally (12); avoid using.
LACTATION: POSSIBLY UNSAFE
when used orally (4); avoid using.
POSSIBLY SAFE ...when used orally and appropriately. Tormentil extract has been used with apparent safety in doses of up to 3 grams daily for up to 3 weeks (12,78590). ...when used topically and appropriately (12).
PREGNANCY AND LACTATION:
Insufficient reliable information available; avoid using.
Below is general information about the interactions of the known ingredients contained in the product Blood Root. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
Below is general information about the adverse effects of the known ingredients contained in the product Blood Root. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
General ...Orally, bloodroot is generally well tolerated when used in appropriate doses, short term (4). Nausea, vomiting, and central nervous system depression have been reported. Higher oral doses can result in glaucoma, hypotension, shock, and coma (6,12). Long term use of bloodroot toothpaste or mouthwash has been associated with leukoplakia, keratoses of the mouth, impaired taste, and staining of the tongue, teeth, and fillings (36666,36668,36707). Topically, skin contact with fresh bloodroot can cause irritation or contact dermatitis (19). Avoid contact with the eyes and mucous membranes because of its irritant properties.
Cardiovascular ...Orally, high doses of bloodroot may cause hypotension (6).
Dermatologic ...Topically, skin contact with fresh bloodroot can cause irritation or contact dermatitis (19). Topical application of bloodroot can corrode skin and produce a thick dry scab called an eschar which falls off and results in a depressed scar. There are numerous cases of patients applying bloodroot salves topically for 3-4 hours daily for 3-12 days to skin cancers, moles and blemishes. These patients report experiencing severe pain, burning, skin erosion, and scarring (53499,95442,95444,95445,95446).
Gastrointestinal ...Orally, bloodroot may cause nausea and vomiting (12). Some research shows that bloodroot-containing toothpastes and mouth rinses can be used for up to six months without evidence of adverse effects (36679). However observational research has found an association between chronic use of bloodroot toothpaste or mouthwash and leukoplakia or keratoses of the mouth and lip. Discontinuing these products did not always result in a resolution of leukoplakia (36666,36668). Prolonged use of bloodroot oral rinse causes impaired sensation of taste, as well as staining of the tongue, teeth, and fillings. The impaired taste and staining do seem to resolve after discontinuation of the bloodroot rinse (36707).
Neurologic/CNS ...Orally, bloodroot may cause CNS depression. High oral doses may cause shock and coma (6).
Ocular/Otic ...There are reports of worsening vision after small oral doses of bloodroot (36680). Glaucoma has been reported after high oral doses of bloodroot (6). However, some experts disagree that oral use of bloodroot causes ocular toxicity (36680). Direct contact of bloodroot with the eyes and mucous membranes should be avoided because of its irritant properties.
General
...Orally, tormentil seems to be well tolerated.
Most Common Adverse Effects:
Orally: Abdominal pain, heartburn, and a feeling of fullness.
Gastrointestinal ...Orally, tormentil has been reported to cause mild abdominal pain, heartburn, and feelings of fullness in one clinical trial. These adverse effects occurred at the same frequency for doses ranging from 1.2 grams daily to 3 grams daily (78590).
